Published online Feb 27, 2024. doi: 10.4254/wjh.v16.i2.241
Peer-review started: October 22, 2023
First decision: December 8, 2023
Revised: December 21, 2023
Accepted: January 8, 2024
Article in press: January 8, 2024
Published online: February 27, 2024
Portal vein thrombosis (PVT) is a commonthsn complication after splenectomy in patients with cirrhosis. However, the predictors of postoperative PVT are not known.
To investigate the predictors of PVT after splenectomy in patient with cirrhosis.
A total of 45 patients with cirrhosis who underwent splenectomy were consecutively enrolled from January 2017 to December 2018. The incidence of PVT at 1 months, 3 months, and 12 months after splenectomy in patients with cirrhosis was observed. The hematological indicators, biochemical and coagulation parameters, and imaging features were recorded at baseline and at each observation point. The univariable, multivariable, receiver operating characteristic curve and time-dependent curve analyses were performed.
The cumulative incidence of PVT was 40.0%, 46.6%, and 48.9% at 1 months, 3 months, and 12 months after splenectomy. Multivariable analysis showed that portal vein diameter (PVD) ≥ 14.5 mm and monthsdel end-stage liver disease (MELD) score > 10 were independent predictors of PVT at 1 months, 3 months, and 12 months after splenectomy (P < 0.05). Time-dependent curve showed that the cumulative incidence of PVT was significantly different between patients with MELD score ≤ 10 and > 10 (P < 0.05). In addition, the cumulative incidence of PVT in the PVD ≥ 14.5 mm group was significantly higher than that in the PVD < 14.5 mm group (P < 0.05).
Wider PVD and MELD score > 10 were independent predictors of PVT at 1 months, 3 months, and 12 months after splenectomy in patient with cirrhosis.
Core Tip: Portal vein diameter (PVD) ≥ 14.5 mm was independent predictors of portal vein thrombosis (PVT) at 1-months, 3-months, and 12-months after splenectomy. End-stage liver disease score > 10 was independent predictors of PVT at 1-months, 3-months, and 12-months after splenectomy. The patients with PVD ≥ 14.5 mm and/or end-stage liver disease score > 10 in preoperative, preoperative treatment of reducing portal vein pressure and improving liver function may help to reduce the incidence of PVT after splenectomy.
- Citation: Li T, Wang LL, Li YP, Gan J, Wei XS, Mao XR, Li JF. Predictors of portal vein thrombosis after splenectomy in patients with cirrhosis. World J Hepatol 2024; 16(2): 241-250
- URL: https://www.wjgnet.com/1948-5182/full/v16/i2/241.htm
- DOI: https://dx.doi.org/10.4254/wjh.v16.i2.241
Portal vein thrombosis (PVT) involves the portal vein and its main branches, and cirrhosis is one of the monthsst commonthsn causes. The natural incidence of PVT in cirrhosis is 3.7%-24.4%[1], and the incidence of advanced cirrhosis is 10%-15%[2]. In cirrhosis, PVT is often latent, and is only discovered accidentally. The treatment of PVT with cirrhosis is controversial.
Currently, splenectomy is one of the main methods of treatment for portal hypertension, hypersplenism and upper gastrointestinal bleeding. Splenectomy can significantly improve the prognosis and survival of patients with cirrhosis[3,4]. Splenectomy decreases portal hypertension, improves liver function, and reduces fibrosis[5-7]. It also improves liver regeneration. A 10-year retrospective follow-up study based on the inverse probability of treatment weighting method found that splenectomy decreased the risk of hepatocellular carcinoma in cirrhosis patients with portal-hypertension-related bleeding[8]. Splenectomy has been considered an effective option to reverse thrombocytopenia in cirrhosis patients with splenomegaly. Thus, splenectomy may be beneficial for treatment of liver cirrhosis with hypersplenism. Splenectomy have been widely used in Asia for the treatment of esophagogastric variceal hemonthsrrhage and hypersplenism caused by cirrhotic portal hypertension. However, splenectomy can increase the risk of PVT at least 10 times[9]. The incidence of PVT was 18.9%-57.0% after splenectomy, which was significantly higher than the natural incidence in patients with cirrhosis without surgery[2,10]. PVT can induce or aggravate upper gastrointestinal bleeding, hepatic encephalopathy, and ascites, increase the risk of intestinal ischemia or intestinal necrosis, reduce the survival of patients and grafts after liver transplantation, and result in chronic cavernous transformation of the portal vein system in the long term[11-14].
Alteration in blood flow, hypercoagulability and vascular endothelial injury are the main risk factors for PVT[2]. PVT is associated with preoperative slower portal vein velocity, wider portal vein diameter (PVD) and splenic vein, and lower preoperative and higher postoperative platelet counts[15-18]. A higher monthsdel end-stage liver disease (MELD) score is associated with hepatic encephalopathy, variceal bleeding, refractory ascites, and spontaneous peritonitis[19-21]. Higher MELD score corresponds with higher monthsrtality in liver transplantation[20]. A higher of MELD score may be associated with postoperative PVT. The mechanisms of PVT after splenectomy are still unclear. Our study aimed to establish the risk factors for PVT after splenectomy and early sensitive indicators, to provide a predictive basis for early PVT.
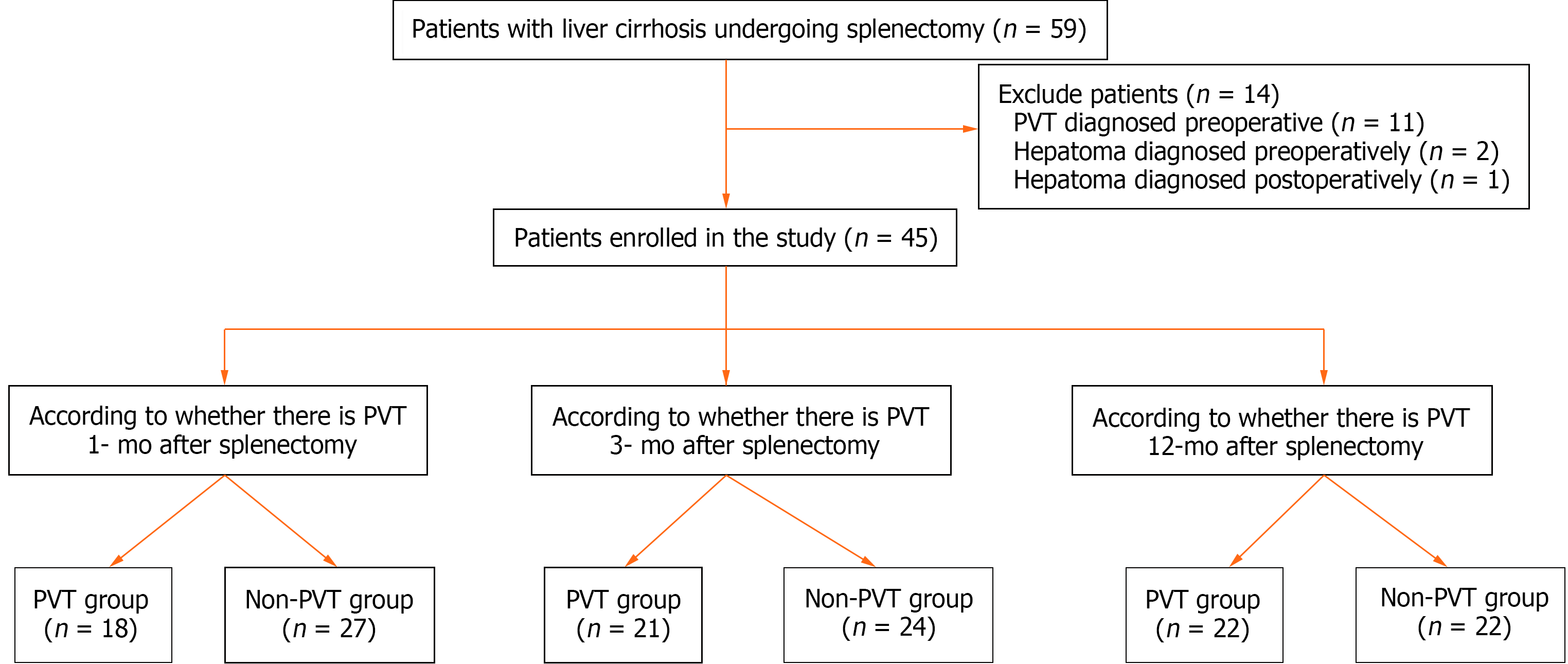
We enrolled 45 consecutive patients with cirrhosis who underwent splenectomy between January 2017 and December 2018 at the First Hospital of Lanzhou University. The flow diagram of the study population is shown in Figure 1. The study was approved by the ethics committee of the first hospital of Lanzhou University (LDYYLL2019-209) and informed consent was obtained from the patients.
Inclusion criteria were histologically proven cirrhosis or cirrhosis diagnosed by a history of liver disease, clinical manifestations, laboratory tests, and imaging studies, and the patients underwent splenectomy. The indications for splenectomy included: Endoscopic treatment-resistant esophagogastric varices with or without variceal hemonthsrrhage; history of esophageal variceal bleeding or potential bleeding; infection caused by hypersplenism and thrombocytopenia (platelet count < 50 × 109/L); and upper abdominal discomfort caused by an enlarged spleen.
The exclusion criteria were as follows: (1) Age > 18 years or < 70 years; (2) patients who developed PVT preoperatively; (3) patients who presented preoperatively with hepatic carcinoma, hepatic encephalopathy, or preoperative Child-Pugh class C, or other tumonthsrs; (4) patients with cirrhosis who underwent liver transplantation; (5) patients who underwent transjugular portal systemic shunt; (6) patients who underwent abdominal surgery; (7) splenectomy for hematological diseases and other reasons (such as trauma); (8) vascular malformation and idiopathic portal hypertension; (9) incomplete clinical data (without hemonthscytes, imaging and other relevant data); (10) coexistence of other serious diseases (shock, multiple organ failure, uremia, and severe infection); and (11) loss to follow-up.
PVT was detected by duplex ultrasonography, computed tomonthsgraphy, or computed tomonthsgraphy angiography. They were performed within 1 wk before the operation to exclude preoperative PVT. Re-examination was performed at 1 months, 3 months, 6 months, and 12 months postoperatively.
Routine blood, parameters and coagulation parameters were measured within 3 d before the operation and used as baseline data. Re-examinations were performed at 1 months, 3 months, 6 months, and 12 months postoperatively in the outpatient or inpatient department of the First Hospital of Lanzhou University. BC-5390 CRP automatic blood cell analyzer (Mindray Bio-Medical Electronics Co. Ltd., Shenzhen, Guangdong Province, China) was used for routine blood testing. The AU400 automatic biochemical analyzer (Olympus Optics Co. Ltd., Japan) was used to detect biochemical parameters. Coagulation parameters were detected by PrecilC3510 automatic coagulation analyzer (Mindray). PVD was measured using Doppler ultrasound (GE Logic E9).
An independent t test or single factor analysis was used to analyze the difference in data in accordance with a normal distribution, and the Mann-Whitney test was used to analyze non-normally distributed data. The χ2 test or Fisher’s exact test was used to analyze categorical variables. A logistic regression monthsdel analyzed the multivariable data. A receiver operating characteristic (ROC) curve was used to evaluate the specificity and sensitivity of PVD and MEDL score for predicting PVT. The Kaplan-Meier method was used to calculate the cumulative incidence of PVT, and the log-rank test was used to compare the difference in the cumulative incidence of PVT between the groups.
We included 45 consecutive patients with cirrhosis who underwent splenectomy (Table 1). The mean age was 47.62 years ± 11.16 years, and 53.3% were female. In terms of etiology, 84% of patients with cirrhosis had hepatitis B, 4.0% hepatitis C, 8.0% autoimmune hepatitis, and 4.0% unexplained cirrhosis. Thirty-five (78.8%) liver cirrhosis patients underwent open splenectomy, and 10 (21.2%) underwent laparoscopic splenectomy.
| Variables | Data |
| Gender | |
| Men, n (%) | 21 (46.7) |
| Female, n (%) | 24 (53.3) |
| Age (yr) | 47.62 ± 11.16 |
| Etiology | |
| HBV, n (%) | 37 (74.0) |
| HCV, n (%) | 2 (4.0) |
| AIH, n (%) | 4 (8.0) |
| Unexplained cirrhosis, n (%) | 2 (4.0) |
| WBC (109/L) | 2.21 ± 1.18 |
| RBC (1012/L) | 3.68 ± 0.78 |
| PLT (109/L) | 44.09 ± 28.54 |
| AST (U/L) | 35.18 ± 14.82 |
| ALT (U/L) | 26.89 ± 14.30 |
| TBIL (μmol/L) | 26.77 ± 15.97 |
| ALB (g/L) | 38.81 ± 4.16 |
| INR | 1.30 ± 0.19 |
| PT (s) | 14.23 ± 1.94 |
| PTA (%) | 68.71 ± 13.45 |
| D-D2 (mg/L) | 0.56 ± 1.03 |
| MELD | 10.76 ± 3.30 |
| PVD (mm) | 13.66 ± 2.49 |
| PV (cm/s) | 19.87 ± 5.88 |
| SVD (mm) | 10.35 ± 2.61 |
| Ascites, n (%) | 19 (42.2) |
| Child-Pugh grade | |
| A, n (%) | 32 (71.1) |
| B, n (%) | 13 (28.9) |
| C, n (%) | 0 (0.0) |
| Operation ways | 1.30 ± 0.19 |
| Open splenectomy, n (%) | 35 (78.8) |
| Laparoscopic splenectomy, n (%) | 10 (21.2) |
| Spleen volume (cm3) | 2349.28 (531.12-13080.00) |
There were 18 (40.0%) patients with PVT and 27 (60.0%) without PVT at 1 months after splenectomy; 21 (46.6%) with PVT and 27 (53.4%) without PVT at 3 months after splenectomy; and 22 (48.9%) with PVT and 27 (61.1%) without PVT at 12 months after the operation.
There were 18 (40.0%) patients with ascites at 1 wk after splenectomy; six (13.3%) patients with ascites at 1 months after splenectomy; one (2.2%) patient with hepatic encephalopathy, four (8.9%) with ascites, and one (2.2%) with upper gastrointestinal hemonthsrrhage at 3 months after splenectomy; one (2.2%) patient with upper gastrointestinal hemonthsrrhage at 6 months after splenectomy; and no decompensation occurred 1 year after the operation. During 1-year follow-up, there was no postoperative monthsrtality.
The demonthsgraphic and laboratory data were compared in patients who developed PVT at 1 months, 3 months, and 12 months postoperatively. Univariable analysis revealed that the incidence of PVT at 1 months, 3 months, and 12 months postoperatively in the MEDL score > 10 group was significantly higher than in the MELD score ≤ 10 group (P < 0.05). Patients with PVT had a wider PVD than those without PVT (P < 0.05). At 3 months postoperatively, the PVT group had a longer prothrombin time (PT) (P < 0.05).
Multivariable analysis identified the following as independent predictors of PVT at 1 months, 3 months, and 12 months postoperatively: Wider preoperative PVD [odds ratio (OR): 2.194, 95% confidence interval (CI): 1.090-4.415, P = 0.028; OR: 1.70, 95%CI: 1.052-2.746, P = 0.030; OR: 1.776, 95%CI: 1.036-3.046, P = 0.037]; and MELD score > 10 (OR: 76.215, 95%CI: 2.534-2287.318, P = 0.013; OR: 12.392, 95%CI: 1.318-116.548, P = 0.028; OR: 23.925, 95%CI: 1.875-305.323, P = 0.015) (Table 2).
| Variable | PVT | Non-PVT | P value | OR | 95%CI |
| Postoperative 1-month | |||||
| Age | 45.72 ± 7.84 | 48.89 ± 12.91 | |||
| PVD | 14.79 ± 2.08 | 12.60 ± 2.38 | 0.028 | 2.194 | 1.090-4.415 |
| MELD > 10 | 12 (70.6) | 8 (32.0) | 0.013 | 76.215 | 2.534-2287.318 |
| PLT | 47.00 ± 41.96 | 42.15 ± 14.78 | |||
| Postoperative 3-months | |||||
| Age | 44.71 ± 7.96 | 44.71 ± 7.96 | |||
| PT | 14.69 ± 1.94 | 14.69 ± 1.94 | |||
| PVD | 14.71 ± 2.16 | 14.71 ± 2.16 | 0.03 | 1.7 | 1.052-2.746 |
| Meld > 10 | 12 (70.6) | 8 (32.0) | 0.028 | 12.392 | 1.318-116.548 |
| PLT | 47.57 ± 38.80 | 41.04 ± 15.11 | |||
| Postoperative 12-months | |||||
| Age | 44.64 ± 7.77 | 50.4 ± 13.19 | |||
| PVD | 14.60 ± 2.13 | 12.64 ± 2.47 | 0.037 | 1.776 | 1.036-3.046 |
| MELD > 10 | 14 (63.6) | 6 (26.1) | 0.015 | 23.925 | 1.875-305.323 |
| PLT | 46.55 ± 38.17 | 41.74 ± 15.05 | |||
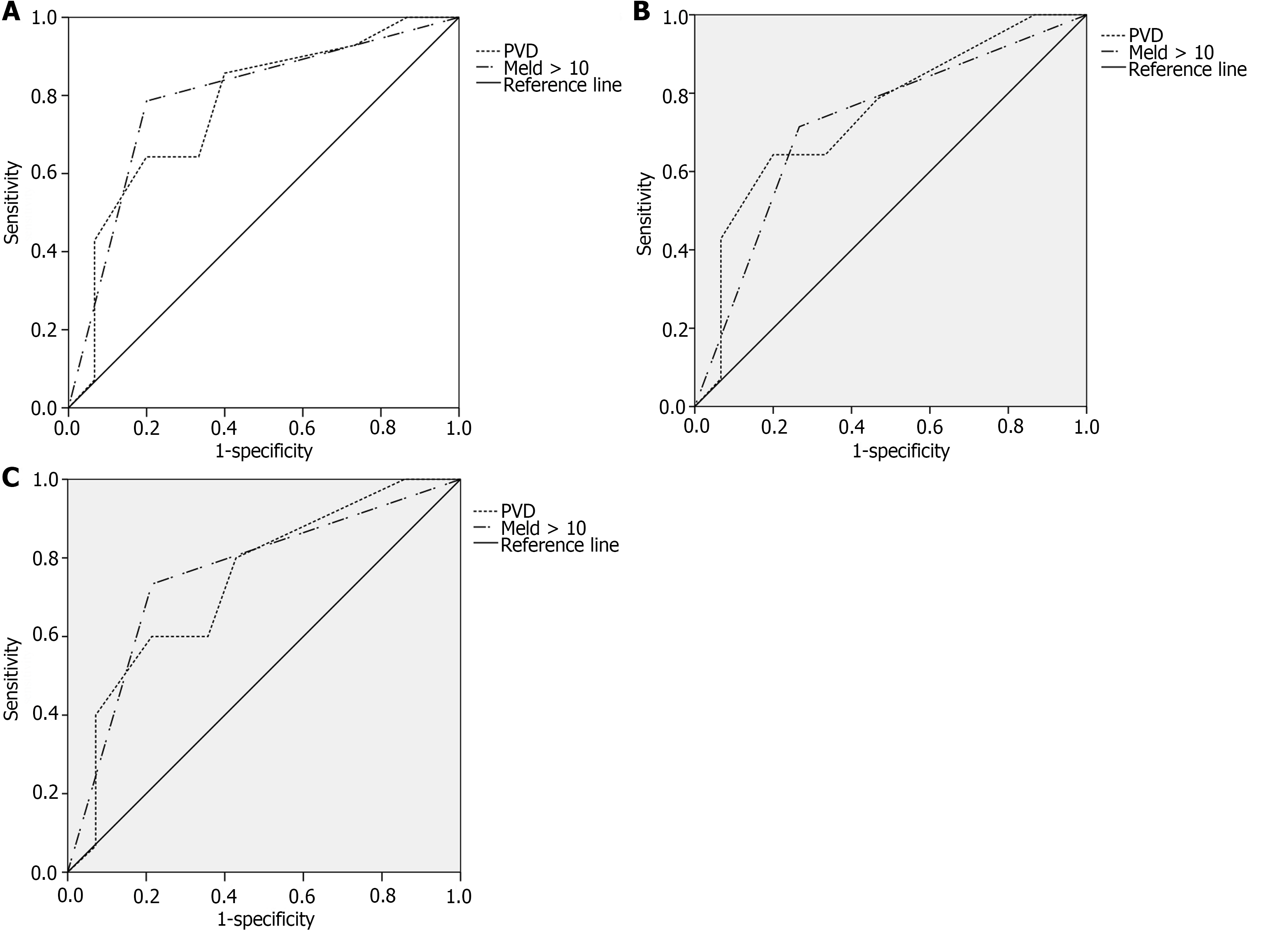
To evaluate the ability of independent predictors to predict PVT after splenectomy, ROC curve analysis was performed (Figure 2). The AUCs of PVD were 0.769, 0.745, and 0.738, respectively (P < 0.05). The AUCs of MELD score > 10 was 0.793, 0.724, and 0.760, respectively (P < 0.05).
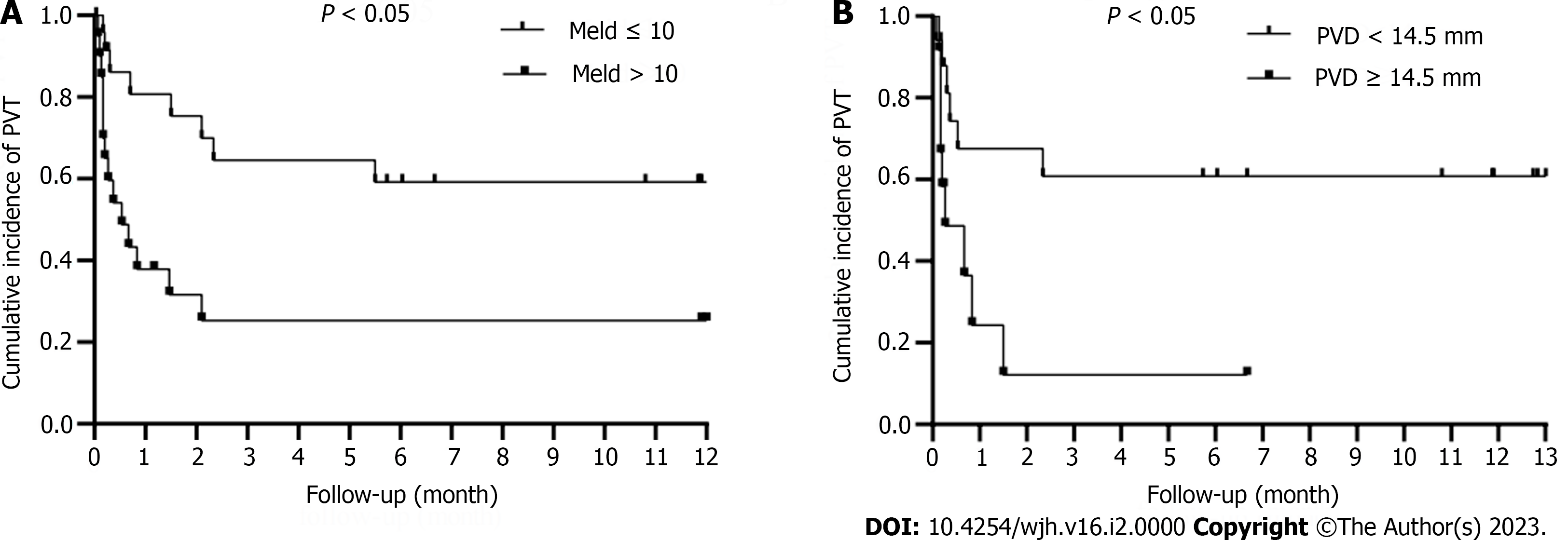
The mean time to occurrence of PVT after splenectomy was 27 d. The optimal cut-off value of PVD was 14.5 mm. The time-dependent curve analysis is shown (Figure 3).
In our observational study, the cumulative incidence of PVT after splenectomy in patients with cirrhosis was 40.0%, 46.6%, 46.6%, and 48.9% at 1 months, 3 months, 6 months, and 12 months, respectively. Wider preoperative PVD and MELD score > 10 may predict the development of PVT after splenectomy. The time-dependent curve analyzed that the development of PVT in patients with MELD score ≤ 10 was lower than in those with MELD score > 10 (P < 0.05). And in the PVD ≥ 14.5 mm group was significantly higher than that in the PVD < 14.5 mm group (P < 0.05).
The cumulative postoperative incidence of PVT was 40.0% at 1 months, 46.6% at 3 months, 46.6% at 6 months, and 48.9% at 12 months. This was similar to the previous study. There are several potential causes of postoperative PVT. Firstly, the occlusion of splenic portal vessels resulted in a reduction in blood flow around the ligation area and enhanced the venous stasis at the splenic vein stump. Secondly, patients with liver cirrhosis are often complicated with changes in blood coagulation proteins, including factor VIII, von Willebrand factor fibrinogen, and tissue factor, putting the blood in a hypercoagulable state[22], which is involved in venous thrombosis. Thirdly, splenectomy can reduce the portal vein flow velocity[23]. The lack of the portal vein flow velocity in our study, we did not obtain a similar conclusion. Previous reports found that wider preoperative splenic vein diameter was an independent predictor of the development of PVT[15,24]. In our study, we found that diameter of the splenic vein in the PVT group was wider than that in the non-PVT group, but there was no significant difference.
Zhang et al[25] considered that the main cause of PVT was the change in portal vein blood flow and not the change in PT or platelet count. We found that a lower preoperative platelet count was not associated with the postoperative development of PVT. In our study, wider PVD was an independent predictor of PVT at 1 months, 3 months, and 12 months after splenectomy. The optimal cut-off value was 14.5 mm. Previous studies reported that PVD > 13.0 mm and > 15.6 mm were independent predictors of PVT after splenectomy[10,26]. Wider PVD means portal hypertension and slower blood flow velocity toward the liver. In addition, a wider PVD can cause a vortex, increase portal vein endothelial cell space, and result in intimal injury and sclerotic changes. The detachment of endothelial cells and the exposure of subintimal collagen fibers activate the endogenous coagulation pathway, increasing the incidence of thrombosis[16,18,27]. Our study provided a favorable indicator for the prediction of PVT after splenectomy.
MELD score is an indicator of the severity of chronic liver disease and the monthsrtality risk of patients with end-stage liver disease. Patients with liver cirrhosis, liver cancer and liver transplantation have different MELD scores, and their prognosis is different[14,28-30]. Previous studies found that a higher MELD score was closely associated with the development of PVT after splenectomy[31]. Our study found that the cumulative incidence of PVT in the MELD score > 10 group was significantly higher than in the MELD score ≤ 10 group. The liver can synthesize coagulation factors and fibrinolytic and antifibrinolytic substances, and inactivate fibrinolysis and antifibrinolytic substances, which play an important role in maintaining the balance of procoagulation and anticoagulation systems. However, the synthesis and inactivation of patients with liver cirrhosis are weakened. Zocco et al[23] found that the reduction in antithrombotic proteins and activation of the hemonthsstatic system were associated with the severity of cirrhosis. Abdel-Razik et al[31] drew the same conclusion. The development of coagulation is associated with the severity of cirrhosis. The balance of the coagulation system in patients with cirrhosis is weak, and this balance is monthsre easily broken after splenectomy. PVT is a marker of portal hypertension and advanced liver cirrhosis, and not a cause. In addition, the MELD score can independently predict PVT recanalization in patients with cirrhosis[32]. The preoperative MELD score can be used as a predictor of postoperative PVT. Preoperative liver function improvement may reduce the incidence of postoperative PVT. Therefore, we should implement splenectomy in patients with good liver function as much as possible.
There were some limitations to our study. Firstly, some patients did not undergo scheduled examinations, and there may have been errors in judgment of the formation time of PVT. Secondly, our study population was small. Thirdly, our study lacked anticoagulation therapy data. However, monthsre prospective, large, randomized studies are needed to assess the risk of development of PVT after splenectomy and provide evidence for anticoagulation therapy.
In conclusion, wider PVD and MELD score > 10 were independent predictors of the development of PVT at 1 months, 3 months, and 12 months after splenectomy in patients with cirrhosis.
Splenectomy has been considered an effective option to reverse thrombocytopenia in cirrhosis patients with splenomegaly. Thus, splenectomy have been widely used in Asia for the treatment of esophagogastric variceal hemonthsrrhage and hypersplenism caused by cirrhotic portal hypertension. However, splenectomy can increase the risk of portal vein thrombosis (PVT) at least 10 times. The incidence of PVT was 18.9%-57.0% after splenectomy, which was significantly higher than the natural incidence in patients with cirrhosis without surgery. PVT can induce or aggravate upper gastrointestinal bleeding, hepatic encephalopathy, and ascites, increase the risk of intestinal ischemia or intestinal necrosis, reduce the survival of patients and grafts after liver transplantation, and result in chronic cavernous transformation of the portal vein system in the long term.
Splenectomy plays an important role in the treatment of cirrhosis. Splenectomy is widely used for the treatment of esophagogastric variceal haemonthsrrhage and hypersplenism owing to cirrhotic portal hypertension. However, splenectomy can increase the risk of PVT at least 10 times. Our study aims to seek the risk factors of PVT after splenectomy and early sensitive indicators, to provide a predictive basis for early PVT and reduce the incidence of PVT.
To establish the risk factors for PVT after splenectomy and early sensitive indicators, to provide a predictive basis for early PVT.
A total of 45 patients with cirrhosis who underwent splenectomy were consecutively enrolled from January 2017 to December 2018. The incidence of PVT at 1 months, 3 months, and 12 months after splenectomy in patients with cirrhosis was observed. The hematological indicators, biochemical and coagulation parameters, and imaging features were recorded at baseline and at each observation point. The univariable, multivariable, receiver operating characteristic curve and time-dependent curve analyses were performed.
PVD ≥ 14.5 mm and monthsdel end-stage liver disease (MELD) > 10 were independent predictors of PVT at 1-months, 3-months, and 12-months after splenectomy. The patients with PVD ≥ 14.5 mm and/or MELD > 10 in preoperative, preoperative treatment of reducing portal vein pressure and improving liver function may help to reduce the incidence of PVT after splenectomy. However, monthsre large-scale studies will be needed to provide reliable and effective evidence for the specific time, drug selection and dosage of anticoagulants.
Portal vein diameter (PVD) ≥ 14.5 mm was independent predictors of PVT at 1-months, 3-months, and 12-months after splenectomy. End-stage liver disease score > 10 was independent predictors of PVT at 1-months, 3-months, and 12-months after splenectomy. The patients with PVD ≥ 14.5mm and/or end-stage liver disease score > 10 in preoperative, preoperative treatment of reducing portal vein pressure and improving liver function may help to reduce the incidence of PVT after splenectomy.
How to prophylactic anticoagulation therapy after splenectomy? Anticoagulant therapy of PVT should be explored.
The authors thank all the participants and all the staff who contributed to this study.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: El-Bendary M, Egypt S-Editor: Chen YL L-Editor: A P-Editor: Cai YX
| 1. | Senzolo M, Garcia-Tsao G, García-Pagán JC. Current knowledge and management of portal vein thrombosis in cirrhosis. J Hepatol. 2021;75:442-453. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 74] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 2. | Intagliata NM, Caldwell SH, Tripodi A. Diagnosis, Development, and Treatment of Portal Vein Thrombosis in Patients With and Without Cirrhosis. Gastroenterology. 2019;156:1582-1599.e1. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 147] [Cited by in F6Publishing: 184] [Article Influence: 36.8] [Reference Citation Analysis (0)] |
| 3. | Ushitora Y, Tashiro H, Takahashi S, Amano H, Oshita A, Kobayashi T, Chayama K, Ohdan H. Splenectomy in chronic hepatic disorders: portal vein thrombosis and improvement of liver function. Dig Surg. 2011;28:9-14. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 58] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 4. | Yang L, Zhang Z, Zheng J, Kong J, Yang X, Wang W. Long-term outcomes of oesophagogastric devascularization and splenectomy in patients with portal hypertension and liver cirrhosis. ANZ J Surg. 2020;90:2269-2273. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 5. | Zhao Y, Zhou R, Guo Y, Chen X, Zhang A, Wang J, Ji F, Qin B, Geng J, Kong G, Li Z. Improvement of gut microbiome and intestinal permeability following splenectomy plus pericardial devascularization in hepatitis B virus-related cirrhotic portal hypertension. Front Immunol. 2022;13:941830. [PubMed] [DOI] [Cited in This Article: ] [Reference Citation Analysis (0)] |
| 6. | Liang QS, Xie JG, Yu C, Feng Z, Ma J, Zhang Y, Wang D, Lu J, Zhuang R, Yin J. Splenectomy improves liver fibrosis via tumonthsr necrosis factor superfamily 14 (LIGHT) through the JNK/TGF-β1 signaling pathway. Exp Monthsl Med. 2021;53:393-406. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 7. | Bai DS, Zhou BH, Qian JJ, Zhang C, Jin SJ, Jiang GQ. Effects of laparoscopic splenectomy and azygoportal disconnection on liver synthesis function and cirrhosis: a 2-year prospective study. Surg Endosc. 2020;34:5074-5082. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7] [Cited by in F6Publishing: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 8. | Gao TM, Zhou J, Xiang XX, Jin SJ, Qian JJ, Zhang C, Zhou BH, Tang H, Bai DS, Jiang GQ. Splenectomy and azygoportal disconnection decreases the risk of hepatocellular carcinoma for cirrhosis patients with portal hypertension bleeding: a 10-year retrospective follow-up study based on the inverse probability of treatment weighting method. J Gastroenterol. 2023;58:503-512. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 2] [Reference Citation Analysis (0)] |
| 9. | Qi X, Han G, Ye C, Zhang Y, Dai J, Peng Y, Deng H, Li J, Hou F, Ning Z, Zhao J, Zhang X, Wang R, Guo X. Splenectomy Causes 10-Fold Increased Risk of Portal Venous System Thrombosis in Liver Cirrhosis Patients. Med Sci Monthsnit. 2016;22:2528-2550. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 10. | Noronha Ferreira C, Marinho RT, Cortez-Pinto H, Ferreira P, Dias MS, Vasconcelos M, Alexandrino P, Serejo F, Pedro AJ, Gonçalves A, Palma S, Leite I, Reis D, Damião F, Valente A, Xavier Brito L, Baldaia C, Fatela N, Ramalho F, Velosa J. Incidence, predictive factors and clinical significance of development of portal vein thrombosis in cirrhosis: A prospective study. Liver Int. 2019;39:1459-1467. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 55] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 11. | Hirashita T, Ohta M, Kai S, Masuda T, Eguchi H, Iwashita Y, Ogawa T, Kitano S. Implications of portal vein thrombosis after splenectomy for patients with idiopathic portal hypertension. Surg Today. 2011;41:1475-1480. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7] [Cited by in F6Publishing: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 12. | Agbim U, Jiang Y, Kedia SK, Singal AK, Ahmed A, Bhamidimarri KR, Bernstein DE, Harrison SA, Younossi ZM, Satapathy SK. Impact of Nonmalignant Portal Vein Thrombosis in Transplant Recipients With Nonalcoholic Steatohepatitis. Liver Transpl. 2019;25:68-78. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 13. | Intagliata NM, Saad WE, Caldwell SH. Effects of restoring portal flow with anticoagulation and partial splenorenal shunt embolization. Hepatology. 2015;61:1088-1090. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 8] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 14. | Berry K, Taylor J, Liou IW, Ioannou GN. Portal vein thrombosis is not associated with increased monthsrtality amonthsng patients with cirrhosis. Clin Gastroenterol Hepatol. 2015;13:585-593. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 61] [Cited by in F6Publishing: 65] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 15. | Kuroki T, Kitasato A, Tokunaga T, Takeshita H, Taniguchi K, Maeda S, Fujioka H. Predictors of portal and splenic vein thrombosis after laparoscopic splenectomy: a retrospective analysis of a single-center experience. Surg Today. 2018;48:804-809. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 16. | Jiang GQ, Bai DS, Chen P, Xia BL, Qian JJ, Jin SJ. Predictors of portal vein system thrombosis after laparoscopic splenectomy and azygoportal disconnection: A Retrospective Cohort Study of 75 Consecutive Patients with 3-monthsnths follow-up. Int J Surg. 2016;30:143-149. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 17. | Li MX, Zhang XF, Liu ZW, Lv Y. Risk factors and clinical characteristics of portal vein thrombosis after splenectomy in patients with liver cirrhosis. Hepatobiliary Pancreat Dis Int. 2013;12:512-519. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 18. | Huang L, Yu Q, Wang J. Association Between Changes in Splanchnic Hemonthsdynamics and Risk Factors of Portal Venous System Thrombosis After Splenectomy with Periesophagogastric Devascularization. Med Sci Monthsnit. 2018;24:4355-4362. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 19. | Obstein KL, Campbell MS, Reddy KR, Yang YX. Association between monthsdel for end-stage liver disease and spontaneous bacterial peritonitis. Am J Gastroenterol. 2007;102:2732-2736. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 46] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 20. | Bertot LC, Gomez EV, Almeida LA, Soler EA, Perez LB. Monthsdel for End-Stage Liver Disease and liver cirrhosis-related complications. Hepatol Int. 2013;7:347-355. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1] [Reference Citation Analysis (0)] |
| 21. | Kamath PS, Kim WR; Advanced Liver Disease Study Group. The monthsdel for end-stage liver disease (MELD). Hepatology. 2007;45:797-805. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1075] [Cited by in F6Publishing: 1118] [Article Influence: 65.8] [Reference Citation Analysis (0)] |
| 22. | Carnevale R, Raparelli V, Nocella C, Bartimonthsccia S, Novo M, Severino A, De Falco E, Cammisotto V, Pasquale C, Crescioli C, Scavalli AS, Riggio O, Basili S, Violi F. Gut-derived endotoxin stimulates factor VIII secretion from endothelial cells. Implications for hypercoagulability in cirrhosis. J Hepatol. 2017;67:950-956. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 62] [Cited by in F6Publishing: 73] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 23. | Zocco MA, Di Stasio E, De Cristofaro R, Novi M, Ainora ME, Ponziani F, Riccardi L, Lancellotti S, Santoliquido A, Flore R, Pompili M, Rapaccini GL, Tondi P, Gasbarrini GB, Landolfi R, Gasbarrini A. Thrombotic risk factors in patients with liver cirrhosis: correlation with MELD scoring system and portal vein thrombosis development. J Hepatol. 2009;51:682-689. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 309] [Cited by in F6Publishing: 337] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 24. | Kinjo N, Kawanaka H, Akahoshi T, Tomikawa M, Yamashita N, Konishi K, Tanoue K, Shirabe K, Hashizume M, Maehara Y. Risk factors for portal venous thrombosis after splenectomy in patients with cirrhosis and portal hypertension. Br J Surg. 2010;97:910-916. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 86] [Cited by in F6Publishing: 97] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 25. | Zhang Y, Wen TF, Yan LN, Yang HJ, Deng XF, Li C, Wang C, Liang GL. Preoperative predictors of portal vein thrombosis after splenectomy with periesophagogastric devascularization. World J Gastroenterol. 2012;18:1834-1839. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 20] [Cited by in F6Publishing: 22] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 26. | Stamonthsu KM, Toutouzas KG, Kekis PB, Nakos S, Gafou A, Manouras A, Krespis E, Katsaragakis S, Bramis J. Prospective study of the incidence and risk factors of postsplenectomy thrombosis of the portal, mesenteric, and splenic veins. Arch Surg. 2006;141:663-669. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 134] [Cited by in F6Publishing: 147] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 27. | Huang D, Tao M, Cao L, Wang X, Zheng S, Cao Y. Risk Factors and Anticoagulation Effects of Portal Vein System Thrombosis After Laparoscopic Splenectomy in Patients With or Without Cirrhosis. Surg Laparosc Endosc Percutan Tech. 2019;29:498-502. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 7] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 28. | Kamath PS, Wiesner RH, Malinchoc M, Kremers W, Therneau TM, Kosberg CL, D’Amico G, Dickson ER, Kim WR. A monthsdel to predict survival in patients with end-stage liver disease. Hepatology. 2001;33:464-470. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3462] [Cited by in F6Publishing: 3434] [Article Influence: 149.3] [Reference Citation Analysis (0)] |
| 29. | Ferlitsch M, Reiberger T, Hoke M, Salzl P, Schwengerer B, Ulbrich G, Payer BA, Trauner M, Peck-Radosavljevic M, Ferlitsch A. von Willebrand factor as new noninvasive predictor of portal hypertension, decompensation and monthsrtality in patients with liver cirrhosis. Hepatology. 2012;56:1439-1447. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 133] [Cited by in F6Publishing: 131] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 30. | Limquiaco JL, Wong GL, Wong VW, Lai PB, Chan HL. Evaluation of monthsdel for end stage liver disease (MELD)-based systems as prognostic index for hepatocellular carcinoma. J Gastroenterol Hepatol. 2009;24:63-69. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 15] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 31. | Abdel-Razik A, Monthsusa N, Elhelaly R, Tawfik A. De-novo portal vein thrombosis in liver cirrhosis: risk factors and correlation with the Monthsdel for End-stage Liver Disease scoring system. Eur J Gastroenterol Hepatol. 2015;27:585-592. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 59] [Cited by in F6Publishing: 61] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 32. | Noronha Ferreira C, Reis D, Cortez-Pinto H, Tato Marinho R, Gonçalves A, Palma S, Leite I, Rodrigues T, Pedro AJ, Alexandrino P, Serejo F, Sobral Dias M, Ferreira P, Vasconcelos M, Damião F, Xavier Brito L, Baldaia C, Fatela N, Ramalho F, Velosa J. Anticoagulation in Cirrhosis and Portal Vein Thrombosis Is Safe and Improves Prognosis in Advanced Cirrhosis. Dig Dis Sci. 2019;64:2671-2683. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |